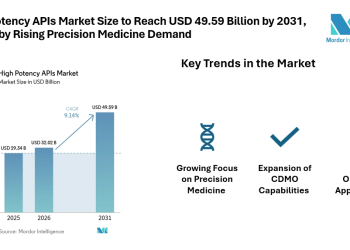
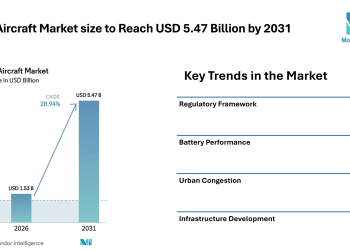
(EMAILWIRE.COM, March 22, 2022 ) Argatroban Market size is estimated to reach $1.65 billion by 2027, growing at a CAGR of 3.9% during the forecast period 2022-2027. Argotroban is a medication termed an anticoagulant or blood thinner. It assists in averting and treating blood clots. In 2000, argatroban was authorized by the Food and Drug Administration (FDA) for prophylaxis or treatment of thrombosis in patients with heparin-induced thrombocytopenia (HIT). Olanzapine is utilized in the treatment of schizophrenia and mania. Thrombocytopenia is discovered amidst communities who take Cellcept, specifically for communities who are male, aged over 60, and who have been taking the medication for less than 1 month. Captopril is an angiotensin-converting enzyme (ACE) inhibitor utilized for the handling of necessary or renovascular hypertension, congestive heart failure, left ventricular dysfunction after myocardial infarction, and nephropathy. The extensive count of heparin-associated complexities amidst patients is set to drive the Argatroban Market. The surging predominance of percutaneous coronary intervention processes and the increased application of argotroban as an anticoagulant is set to propel the growth of the Argatroban Market during the forecast period 2022-2027. This represents the Argatroban Industry Outlook.
Argatroban Market Segment Analysis By Product:
The Argatroban Market based on the product can be further segmented into Argatroban Powder and Argatroban Injection. The Argatroban Powder Segment held the largest market share in 2021. This growth is owing to the surging application of argatroban powder as a raw material of anticoagulant medication for the treatment and aversion of blood clots for patients with heparin-induced thrombocytopenia. The ease of availability online is further propelling the growth of the Argatroban Powder segment.
Furthermore, the Argatroban Injection segment is estimated to grow with the fastest CAGR of 4.6% during the forecast period 2022-2027 owing to the heightening application of Acova (Argatrobin) which is available as a solution for injection to be executed by a healthcare provider into the vein as an anticoagulant for treating thrombosis in patients with heparin-induced thrombocytopenia.
Argatroban Market Segment Analysis By Application:
The Argatroban Market based on the application can be further segmented into Heparin-Induced Thrombocytopenia, Percutaneous Coronary Intervention, and Cerebral Arterial Thrombosis. The Heparin-Induced Thrombocytopenia Segment held the largest market share in 2021. This growth is owing to the surging application of argatroban therapy to treat heparin-Induced thrombocytopenia resulting in considerable minimization of novel thrombosis. The augmented safety and efficiency of argatroban as an agent for the heparin-Induced thrombocytopenia indication is further propelling the growth of this segment.
Furthermore, the Percutaneous Coronary Intervention segment is estimated to grow with the fastest CAGR of 4.7% during the forecast period 2022-2027 owing to the expanded utilization of argatroban for prophylaxis at the time of percutaneous coronary intervention apart from being a substitute anticoagulant in the place of heparin for treatment of heparin-induced thrombocytopenia.
Request for Sample Report @ https://www.industryarc.com/pdfdownload.php?id=501906
Report Price: $ 4500 (Single User License)
Argatroban Market Segment Analysis By Geography:
The Argatroban Market based on geography can be further segmented into North America, Europe, Asia-Pacific, South America, and the Rest of the World. North American Argatroban Market held the largest share with 39% of the overall market in 2021. The growth of this region is owing to the extensive application of argatroban in different applications like heparin-induced thrombocytopenia. The growing application of argatroban as efficient substitute anticoagulation in patients with or at hazard for heparin-induced thrombocytopenia during renal replacement therapy is further propelling the growth of the Argatroban Market in the North American region.
Furthermore, the Asia-Pacific region is estimated to be the region with the fastest CAGR rate over the forecast period 2022-2027. This growth is owing to factors like the surging count of heparin-associated complexities like heparin-induced thrombocytopenia amidst patients. The extensive application of argatroban in the treatment of cerebral arterial thrombosis in the region is further fuelling the progress of the Argatroban Market in the Asia-Pacific region.
Argatroban Market Drivers
Increased Application Of Argatroban In Heparin-Induced Thrombocytopenia (HIT) Is Projected To Drive The Growth Of Argatroban Market:
Heparin-induced thrombocytopenia (HIT) is a severe, life-alarming complexity that takes place in 1-3% of patients taking heparin. Patients with untreated HIT have an up to 50% hazard of establishing life- and limb-alarming thromboembolic complexities. Treatment is based on clinical distrust, halting heparin therapy, and introduction of anticoagulation with a very quickly acting substitute non-heparin anticoagulant, like argatroban-a hepatically excreted direct thrombin inhibitor which is efficient in the treatment of HIT. Argatroban is a secure and efficient treatment for HIT. In patients consuming other hepatically cleared medicines, more reduced initial doses may have to be utilized to avert over-anticoagulation. Clinical and laboratory findings have resulted in most clinicians deducing that patients with HIT should be treated by the discontinuance of heparin therapy and accelerated initiation of a substitute anticoagulant that does not mirror heparin or react with HIT antibodies. The extensive application of argatroban in the treatment of Heparin-induced thrombocytopenia (HIT) is fuelling the growth of the Argatroban Market during the forecast period 2022-2027.
Increased Endorsement Of Argatroban For Treatment Of Different Applications Is Expected To Boost The Demand Of Argatroban:
Argatroban is a direct thrombin inhibitor discovered by Shosuke Okamoto in Japan in the 1970s. Argatroban was utilized clinically earliest in Japan in patients for treatment of peripheral arterial occlusive disease in the initial part of the 1980s. Presently it is endorsed in Japan for treatment of arterial thrombosis (1990), acute cerebral thrombosis (1996), and anticoagulation of AT-deficient patients enduring hemodialysis (1996). Argatroban is also endorsed in Korea for application in chronic arterial occlusion and acute cerebral thrombosis. Argatroban was endorsed in the U.S. in 2000 for both prophylaxis and treatment of thrombosis related to heparin-induced thrombocytopenia (HIT) type II. In Canada, argatroban is endorsed as an anticoagulant for HIT patients who, from the point of view of the accompanying physician, need anticoagulation. A submission to the European health authorities for the application of argatroban in patients with HIT type II is in process. The proliferating endorsement of argatroban for the treatment of different applications is driving the growth of the Argatroban Market during the forecast period 2022-2027.
Direct Purchase link @ https://www.industryarc.com/purchasereport.php?id=501906
Argatroban Market Challenges
Issues Faced With Argatroban Anticoagulation During Cardiopulmonary Bypass Present Challenges To The Growth Of The Argatroban Market:
The application of argatroban as a substitute to heparin during cardiopulmonary bypass (CPB) in patients with heparin-induced thrombocytopenia has acquired attention in the past twenty years. Dosing of argatroban at the time of CPB is complicated owing to a dearth of total comprehension of its pharmacokinetic profile and the different elements during CPB that may change its plasma levels. There is a case where the issues in dosing argatroban resulted in failure to offer sufficient anticoagulation during CPB, as demonstrated by clot formation in the oxygenator, and considerable bleeding in the postoperative period. Some limitations of argatroban detected were the slow commencement of action (30 min), a deferred peak impact (nearly 2 h), and the dearth of a particular antidote. Argatrobans short half-life calls for its execution as a continuous intravenous infusion to generate the anticoagulant impact required at the time of CPB. Argatroban has been utilized in keeping up extracorporeal support not only in adults, but in children, babies, and even neonates. At the time of CPB utilizing argatroban, there have been issues with insufficient anticoagulation leading to clotting of the extracorporeal circuit and at the same time anxiety regarding raised bleeding in the postoperative period owing to the latency in normalization of the Activated Clotting Time (ACT). Clots in the oxygenator have been noted in the post bypass period after discontinuance of argatroban, but more confusing is the reality that people have noted clot formation even during CPB at an ACT of 495 s. This kind of issue is hampering the growth of the Argatroban Market.
Argatroban Industry Outlook
Product launches, mergers and acquisitions, joint ventures, and R&D activities are key strategies adopted by players in this market. Argatroban top 10 companies include:
Pfizer
GlaxoSmithKline
SANDOZ
Eagle Pharmaceuticals
Fresenius Kabi
Teva Pharmaceutical Industries
Mitsubishi Chemical
WEST-WARD
ChemWerth
Gland Pharma
Acquisitions/Product Launches:
In November 2021, Pfizer Inc. has endorsed a deal with a U.N.-supported group to permit additional makers to prepare its experimental VOID-19 pill, a move that could make the treatment accessible to over half of the global population. In a statement circulated, Pfizer pronounced that it would authorize a license for the antiviral pill to the Geneva-based Medicines Patent Pool, which would allow generic drug firms to manufacture the pill for application in 95 countries, constituting around 53% of the global population. The deal rules out certain big countries that have endured overwhelming coronavirus epidemics.
In July 2021, Fresenius Kabi has included a fentanyl product in its series of ready-to-dispense prefilled syringes. The 1-mL presentation, which Fresenius communicated is rare on the U.S. market, is planned to cut waste and diversion by minimizing the possibility for errors. Like other products in Fresenius Simplist series, the fentanyl syringe needs no assembly.
In October 2020, Sandoz Inc declared that its earliest three injectable medications with Radio Frequency Identification (RFID) tags will be accessible this month to U.S. hospitals by way of its collaboration with Kit Check, Inc. The earliest three medications are inclusive of Anectine 200 mg/10mL and Rocuronium 50mg/5mL and 10mg/5mL (paralytics to recline muscles at the time of surgery or additional medical processes), and Norepinephrine 4mg/4mL (to raise/keep up blood pressure). Numerous Sandoz medicines are estimated to be made accessible to Kit Checks hospital partners through early 2021.
Key Takeaways
Geographically, North America Argatroban Market accounted for the highest revenue share in 2021 and it is poised to dominate the market over the period 2022-2027 owing to the application of different dosages of anticoagulant termed argotroban for mature grown-up patients with heparin-induced thrombocytopenia with varying degrees of hepatic impairment in the North American region.
Argatroban Market growth is being driven by the increasing application of anticoagulant termed argatroban to treat or avert blood clots in mature grownups who have thrombocytopenia (reduced levels of platelets in the blood) brought about by utilizing heparin. However, the principal side effect of argatroban is bleeding which can be handled only by halting the argatroban infusion and offering supporting therapy and this is one of the major factors hampering the growth of the Argatroban Market.
Argatroban Market Detailed Analysis on the Strength, Weakness, and Opportunities of the prominent players operating in the market will be provided in the Argatroban Market report.
Related Report:
Heparin Market
https://www.industryarc.com/Report/17362/heparin-market.html
For more Lifesciences and Healthcare related reports, please click here
About IndustryARC: IndustryARC primarily focuses on Cutting Edge Technologies and Newer Applications market research. Our Custom Research Services are designed to provide insights on the constant flux in the global supply-demand gap of markets. Our strong team of analysts enables us to meet the client research needs at a rapid speed, with a variety of options for your business. Any other custom requirements can be discussed with our team, drop an e-mail to sales@industryarc.com to discuss more about our consulting services.