(EMAILWIRE.COM, April 30, 2022 ) The CAR T-Cell Therapy Market is all set to create greater strides in the upcoming period. Big data analytics is flowing through the veins of the healthcare vertical these days. This practice helps in providing better insights to healthcare practitioners. The healthcare vertical has started depending on analytics expertise for deciphering data collected through various sources and monitoring processes efficiency.
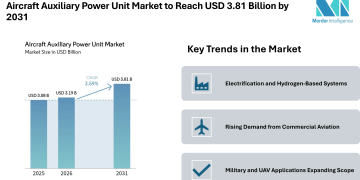
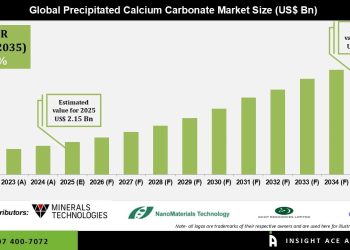
The global CAR T-cell therapy market is anticipated to experience high growth over the coming years. The market is expected to grow from US$ 1.2 Bn in 2020 to over US$ 2.8 Bn by 2031, which reflects a CAGR of around 5.6% for the next ten years.
Key players in the CAR T-cell therapy space are focusing on acquisitions and collaborations with other players of the market to strengthen their position by offering a wide range of products.
For instance, in December 2017, Gilead Science acquired Cell Design Labs Inc., which is a privately held company of biotherapeutics that discovers and develops cell-based therapies. This acquisition benefits Gilead through gaining novel technology platforms that will its boost R&D efforts in cellular therapy.
Get Sample Copy of this Report@ https://www.persistencemarketresearch.com/samples/28264
Also, increasing number of clinical trials in the field of life sciences and CAR T-cells will benefit manufacturers and researchers in developing innovative cancer therapy. With a large extent of studies taking place around CAR T-cell therapy, it is anticipated to enhance product availability in the market over the coming years.
For instance, the experiment on AntiCD19 Chimeric Antigen Receptor T Cells for the treatment of relapsed or refractory Non-Hodgkin lymphoma by Benjamin Tomlinson is undergoing and is currently in phase I clinical trial.
Another study by Janssen Research & Development, LLC of a chimeric antigen receptor T Cell (CAR-T) therapy for myeloma is in phase 2 clinical trial.
Furthermore, a study on the safety and efficacy of ALLO-605 in patients with relapsed/refractory multiple myeloma by Allogene Therapeutics is currently in phase 2.
All these factors will drive demand for CAR T-cell therapy during the forecast period (2021 to 2031).
Company Profiles:
Novartis AG
Gilead Sciences Inc.
Celgene Corporation
bluebird bio, Inc.
Request for Methodology@ https://www.persistencemarketresearch.com/methodology/28264
Key Takeaways from Market Study
By product, Yescarta (axicabtagene ciloleucel) holds high share of approximately 59%.
By indication, relapsed large B-cell lymphoma accounts for approximately 93% market share.
By end user, cancer treatment centers account for around 92% share in the market.
By region, North America holds the largest share of 68.1% of the global CAR T-cell therapy market, due to increasing number of life science companies focusing on the development and commercial manufacturing of CAR T-cells in the region.
Increasing prevalence of cancer and continuous R&D on cell-based therapies are expected to drive demand for CAR T-cell therapy over the decade, says a PMR analyst.
Who is Winning in This Space?
Many of the chief players in the CAR T-cell therapy market are focusing on product launches and collaborations for global expansion.
In March 2018, Celgene Corporation completed the acquisition of Juno Therapeutics, Inc. This acquisition helps Celgene advance in cellular immunotherapy by adding novel technology and advanced cellular manufacturing capabilities to its portfolio.
In August 2017, Gilead Science announced the acquisition of Kite Pharma Inc., an industry leader in the emerging field of cell therapy.
In March 2021, Kite Pharma gained FDA approval for axicabtagene ciloleucel (axi-cel), a CAR T-cell based therapy.
Access Full Report@ https://www.persistencemarketresearch.com/checkout/28264
Want More Insights?
PMR brings the comprehensive research report on forecasted revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2016 to 2031.
The market is segmented in detail based on product (Yescarta (axicabtagene ciloleucel), Kymriah (tisagenlecleucel), JCAR017 (lisocabtagene maraleucel), and bb2121) indication (Relapsed Large B-cell Lymphoma, Acute Lymphoblastic Leukemia (ALL), and Multiple Myeloma), and end user (hospitals and cancer treatment centers), across seven major regions of the world.
Related Reports:
Immune Checkpoint Inhibitors Market – Expanding at a booming CAGR of 14.6%, the global market for immune checkpoint inhibitors market is expected to reach a value beyond US$ 35 Bn over 2018-2026.
Meningococcal Vaccines Market – The meningococcal vaccine market report also projects a significant growth prospective with a projected average Y-o-Y growth rate of ~ 10% during the forecast period.
About Us: Persistence Market Research